FIX-KG-13
WRIST WRAP
Pediatric wrist wrap FIX-KG-13 is designed to offer support for the wrist. The wrist support maintains the constant temperature, reduces pain and improves healing.
Special features
 | SHOULDER BRACE |
 | 1st CLASS MEDICAL PRODUCT |
 | 24/7 THERAPY |
 | WATERPROOF |
 | ALSO AVAILABLE IN VERSION FOR INFANTS |
 | BREATHABLE |
 | UNIVERSAL SIZE |
 | DOUBLE-SIDED |
Indications
- wrist sprain
- wrist inflammation
- overused wrist joints
- joint capsule injury
- wrist RA
How does it work?
Pediatric wrist brace FIX-KG-13 is made of thin, colorful neoprene which provides constant compression and warmth of CMC joint. Wrap around design of FIX-KG-13 provides warmth and smooth circumference adjustment. Due to this fact, our wrist orthosis maintains the temperature, reduces the pain and improves the healing. The pediatric brace FIX-KG-13 is the best option of treatment in case of wrist strain.
ActivePren™ is an active three-layer material consisting of two elastic jersey cover fabrics and a core made of neoprene foam. This material is characterized by softness and high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of ActivePren are the strongest and most effective stabilizing orthoses available on the market.
Available sizes
| Size | Wrist circumference | How to measure |
|---|---|---|
| Universal | min 9 cm – max 15 cm | 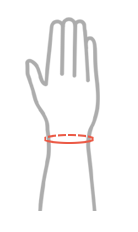 |
Fits for both wrist.
Total height of the product: 5 cm
The product is sent in a random color.

Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.




 Class I medical device in accordance with the Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.
Class I medical device in accordance with the Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.